ASUS MH Series Clinical Displays Listed as Class 1 Devices by US FDA
Mar 04, 2024
All three monitors meet the DICOM Part 14 Grayscale Standard Display Function (GSDF) AAPM TG270 standard, and each includes a scheduled calibration feature, dynamic brightness compensation, and dynamic DICOM compensation to ensure consistent and highly accurate medical images. The monitors feature an anti-glare, low-reflection (AGLR) coating on the panel, and have undergone stringent performance tests. They have been certified by TÜV Rheinland to ensure flicker-free performance and low blue light emissions. These clinical displays also meet ISO 13485 requirements, and are certified for use in clinics and large medical institutions.
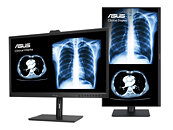
In addition to FDA certification, ASUS is seeking CE marks indicating compliance with EU Medical Device Regulation guidelines as well as approval from the Taiwan Food and Drug Administration (TFDA) for its clinical displays. Aiming to provide a range of new solutions for European and Asia markets, ASUS is also actively pursuing partnerships with major medical institutions across the globe, seeking to better understand industry needs and to respond to ever-changing medical-device market.


View at TechPowerUp Main Site